
Personalized, targeted treatment approaches that harness the body's own immune cells to fight cancer.
Explore Treatment PlatformsCell therapy is a next-generation treatment approach that uses the patient's own immune cells or donor-derived cells to combat disease, particularly cancer. Unlike conventional chemotherapy and radiation, it destroys only target cells without harming healthy tissue.
The CAR-T era began in 2017 with the FDA approval of Kymriah. By early 2024, it had grown to six products: Kymriah, Yescarta, Tecartus, Breyanzi, Abecma, and Carvykti.
These therapies are used in the following cancers:
• Childhood leukemia (B-ALL)
• Large B-cell lymphoma
• Mantle cell lymphoma
• Multiple myeloma (bone marrow cancer)
By late 2024, two new approvals reached solid tumors as well: AMTAGVI (for melanoma) and TECELRA (for synovial sarcoma).
Bottom line: Cell therapy is no longer experimental. It has become a regulatory-approved, evidence-based treatment standard.
CAR-T cell therapy response rates vary by cancer type. Results from two pivotal trials:
• Pediatric leukemia (B-ALL): 81% complete remission in the ELIANA trial (Maude et al., NEJM 2018)
• Refractory large B-cell lymphoma: 83% overall response and 58% complete response in the ZUMA-1 trial (Neelapu et al., NEJM 2017)
• 5-year follow-up: 42.6% of patients still alive
Bottom line: These rates show that in refractory cases where conventional chemotherapy and radiation have failed, cell therapy is a life-saving option.
The global cell therapy market is growing rapidly:
• 2025 valuation: $7-18 billion (varies by research firm)
• 2034 projection: expected to exceed $47 billion
• Annual growth: above 20%
Three forces drive this growth: expansion of CAR-T therapies into new cancer types, declining manufacturing costs, and increasing patient access in emerging markets.
Bottom line: With cGMP-compliant domestic manufacturing in Türkiye, Genkord Therapeutics holds a strategic position in this rapidly growing global market.
Genkord Therapeutics manufactures three ministry-approved cellular therapy platforms in its own cGMP-certified laboratory.
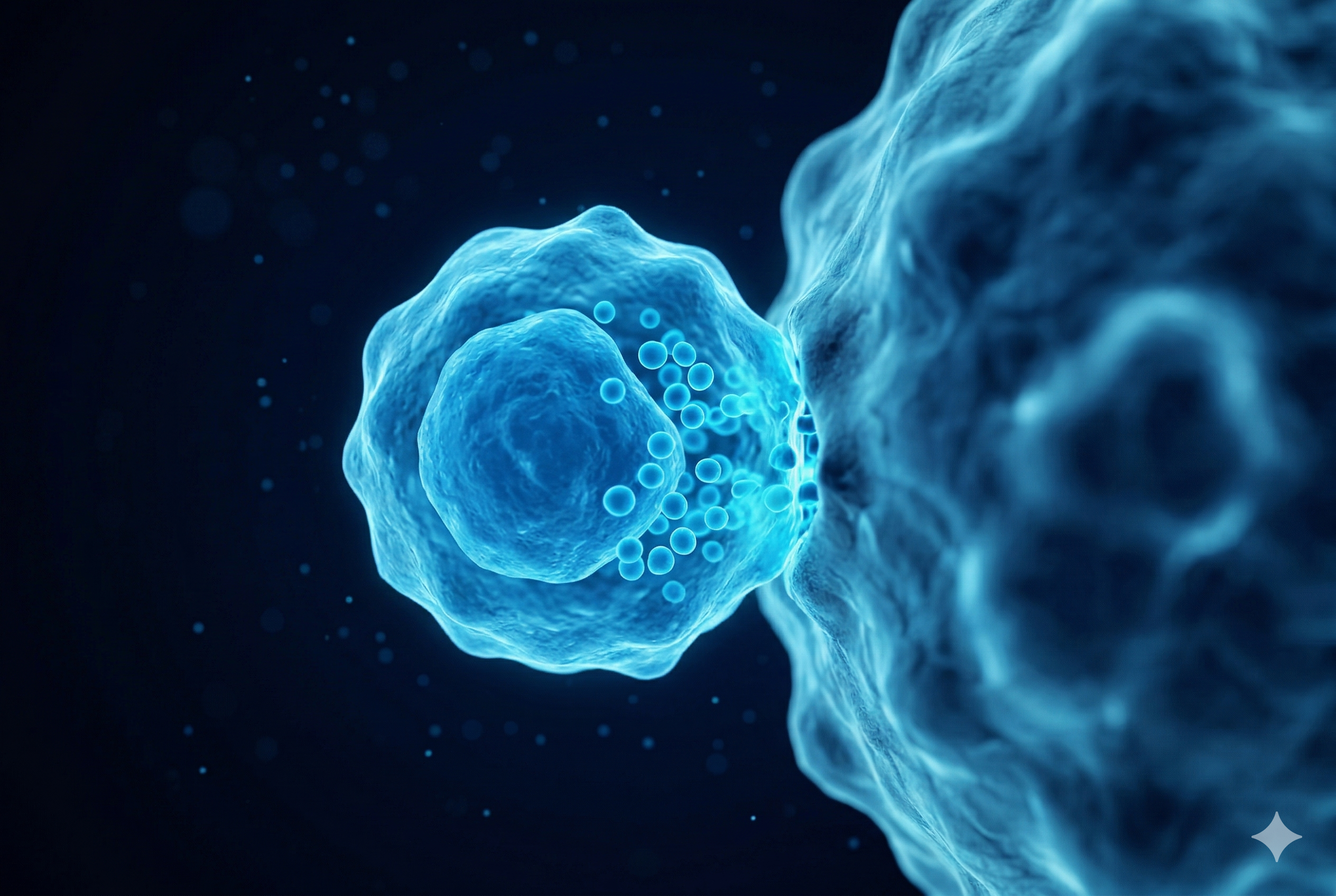
Natural Killer Cell
The elite warriors of innate immunity. Capable of recognizing and destroying tumor cells instantly without prior sensitization. A safe treatment option with a favorable side-effect profile.
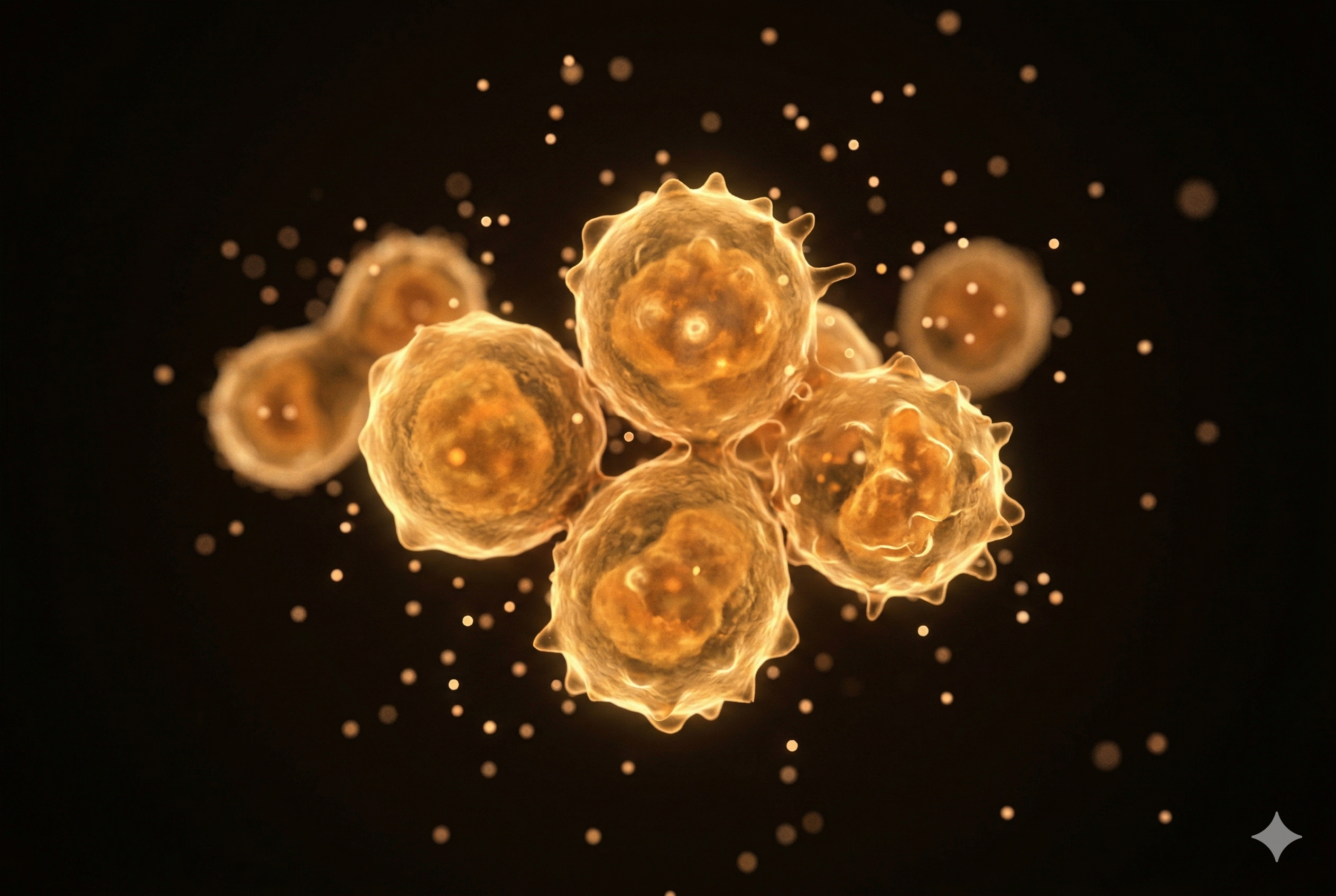
Cytokine-Induced Killer Cell
Cytokine-activated killer cells combining NK and T cell properties. Their hybrid phenotype enables broad-spectrum anti-tumor activity with MHC-unrestricted target recognition.
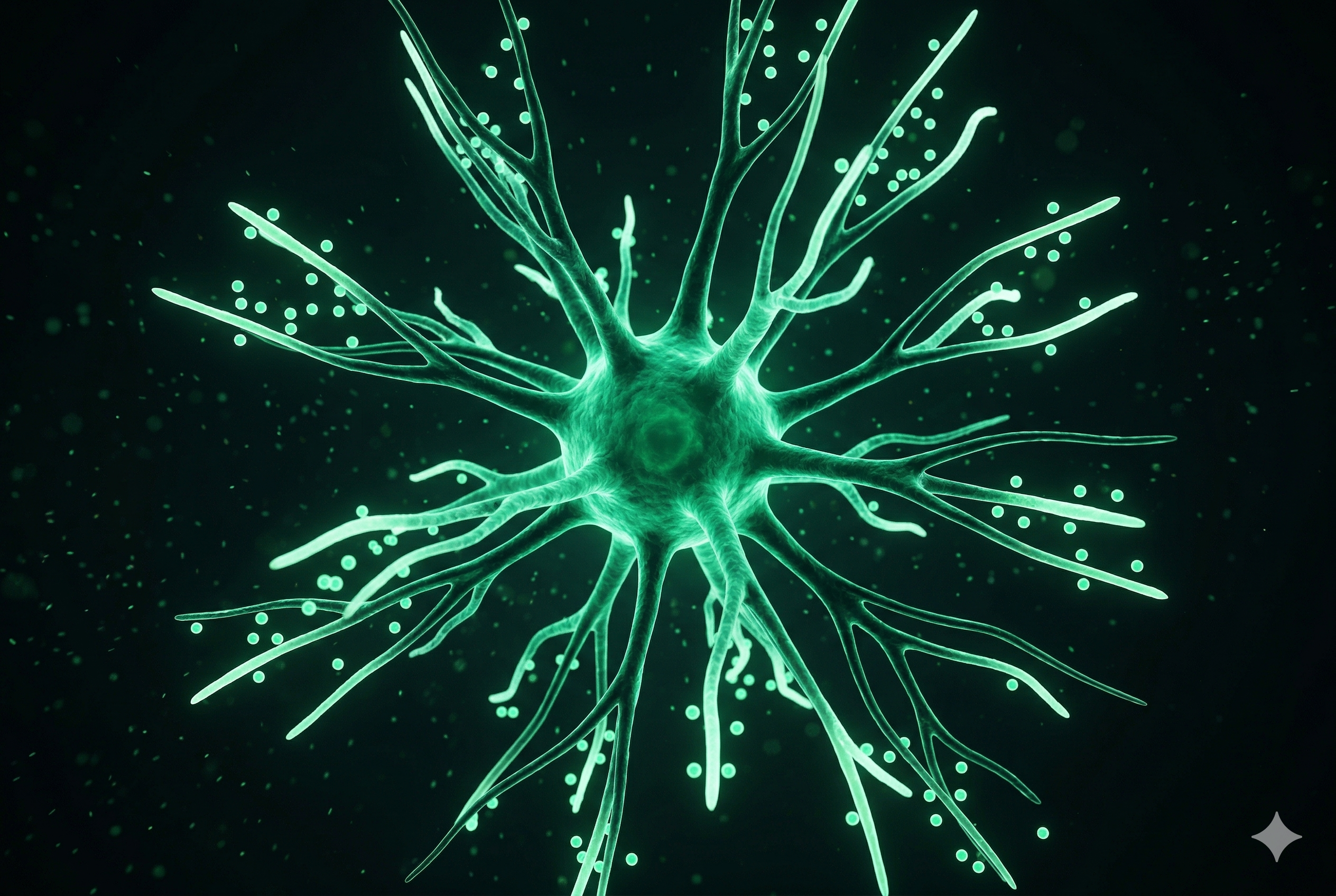
Dendritic Cell
The "teacher" cells of the immune system. They process and present tumor antigens to T cells, triggering a potent, targeted immune response. The foundation of personalized cancer vaccine approaches.

Operating in cell biology since 2003, Genkord develops and manufactures ministry-approved cellular immunotherapy products in its own cGMP-certified R&D facility.
All processes from cell isolation to final product are carried out under one roof in our cGMP-certified R&D facility.
Over 20 years of cell biology expertise. More than 15 biological products successfully developed and commercialized.
NK cell, CIK, and Dendritic Cell: three distinct ministry-approved cellular therapy platforms actively in production.


Get in touch with our team for R&D collaborations, clinical study partnerships, or product information.